Copper crystallizes in a face-centered cubic lattice (the Cu atoms are at the lattice points and at the face centers). If the density of the metal is 8.96 g/cm^3, what is the

SOLVED:Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g/cm3, a radius of 138 pm, and a molar mass of 106.42 g/mol. Use these data to calculate Avogadro's

Chapter 3 Homework - Chapter 3 Homework Textbook Problems 9 17 22 25 33 40 44 46 71 79 Additional problems and solutions Problem#1 Palladium | Course Hero
Problem #1: Palladium crystallizes in a face-centered cubic unit cell Its density is 12023 g/cm 3 Calculate the atomic radius of palladium | Course Hero

Copper crystallizes in a cubic structure. If the density of the metal is 8.% g/cm^3 and the length of the unit cell edge is 361 picometers, find the number of atoms in

SOLVED:Metallic iridium crystallizes in a face-centered cubic lattice, with one Ir atom per lattice point. If the edge length of the unit cell is found to be 382 pm, what is the
If the radius of palladium is 248 pm and the lattice type is body centered cubic, what is the - Sarthaks eConnect | Largest Online Education Community

SOLVED:Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g/cm3, a radius of 138 pm, and a molar mass of 106.42 g/mol. Use these data to calculate Avogadro's

Document - Problem#1 Palladium crystallizes in a face-centered cubic unit cell Its density is 12.023 g\/cm3 Calculate the atomic radius of palladium | Course Hero

SOLVED:**30) Gallium crystallizes in primititre cubic Unit cell The length of the unit cell edge is 3.70 4 The radius ofa Ga atom is 4)7.40 B) 3.70C) 1.85D) 0.930 E) Insufficient data
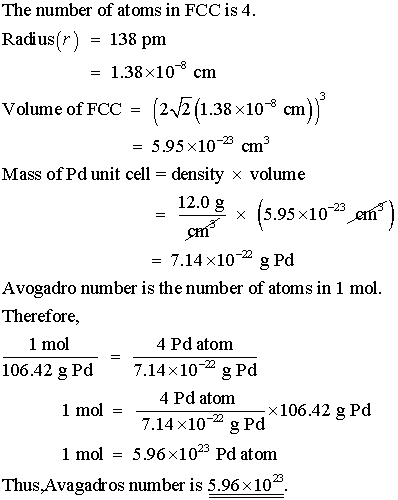
OneClass: Palladium crystallizes with a face-centered cubic structure. It hasa density of 12.0 g/cm3,...
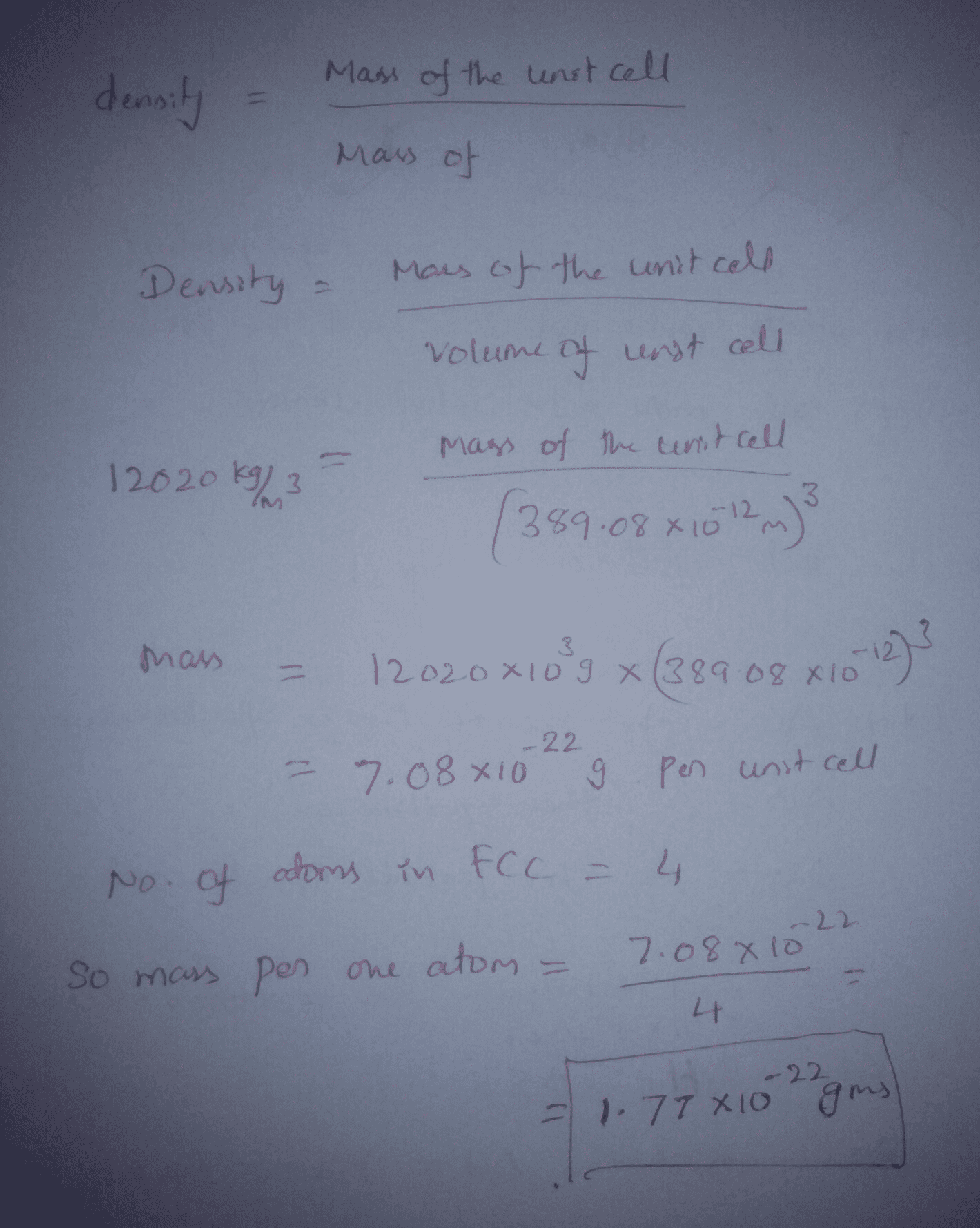
OneClass: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is ...

SOLVED:16. Palladium crystallizes in a face-centered cubic unit cell. Its density is 12.0 glem'. What is the atomic radius of Pd? A. 138 pm B. 1.95 * 10 8 nm C.1.95 * 10-8 cm D. 154 pm E. 0.109 nm

Problem #1: Palladium crystallizes in a face-centered cubic unit cell Its density is 12023 g/cm 3 Calculate the atomic radius of palladium | Course Hero